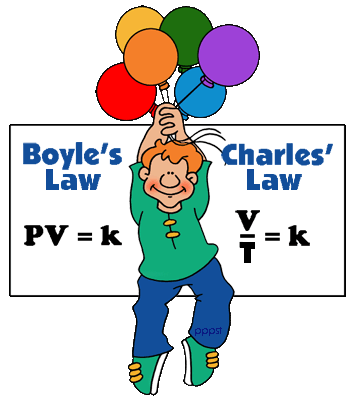GAS LAWS
One of the most amazing things about gases is that, despite wide differences in chemical properties, all the gases more or less obey the gas laws. The gas laws deal with how gases behave with respect to pressure, volume, temperature, and amount.
Contents
Boyle's law:
Boyle's law shows that, at constant temperature, the product of an ideal gas pressure and volume is always constant. It can be determined experimentally using a pressure gauge and a variable volume container. It can also be found through the use of logic; if a container, with a fixed number of molecules inside, is reduced in volume, more molecules will hit the sides of the container per unit time, causing a greater pressure.
As a mathematical equation, Boyle's law is:
P1V1 = P2V2
where P is the pressure (Pa), V the volume (m3) of a gas, and k1 is the constant from this equation—it is not the same as the constants from the other equations below.
This is known as Boyle's law which states: the volume of a given mass of gas is inversely proportional to its pressure, if the temperature remains constant. Mathematically this is:
V= K/P
(k is a constant)
Charle's law:
Charles's Law is an ideal gas law where at constant pressure, the volume of an ideal gas is directly proportional to its absolute temperature.
V1/T1 = Vf/Tf
where:
V1 = initial pressure
T1 = initial temperature
Vf = final pressure
Tf = final temperature
Gay-Lussac's law:
Gives the relationship between pressure and temperature when volume and amount are held constant.
If the temperature of a container is increased, the pressure increases.
If the temperature of a container is decreased, the pressure decreases.
The mathematical form of Gay-Lussac's Law is: P ÷ T = k
This means that the pressure-temperature fraction will always be the same value if the volume and amount remain constant.
Let P1 and T1 be a pressure-temperature pair of data at the start of an experiment. If the temperature is changed to a new value called T2, then the pressure will change to P2.
This equation of P1 ÷ T1 = P2 ÷ T2 will be very helpful in solving Gay-Lussac's Law problems.
Example: 5.00 L of a gas is collected at 22.0°C and 745.0 mmHg. When the temperature is changed to standard, what is the new pressure?
Solution: convert to Kelvin and insert:
745.0 mmHg / 295.0 K = x/ 273.0 K
Cross multiply and divide for the new pressure.
Avogadro's laws:
Avogadro's law is an experimental gas law relating volume of a gas to the amount of substance of gas present. A modern statement of Avogadro's law is:
A law that states, equal volumes of all gases, at the same temperature and pressure, have the same number of molecules.
For a given mass of an ideal gas, the volume and amount (moles) of the gas are directly proportional if the temperature and pressure are constant.
which can be written as:
v/n = K
The most important consequence of Avogadro's law is that the ideal gas constant has the same value for all gases. This means that the constant
P1 x V1 / T1x n1 = P2 x V2 / T2 x n2 = constant
Example:
A 6.0 L sample at 25 °C and 2.00 atm of pressure contains 0.5 moles of a gas. If an additional 0.25 moles of gas at the same pressure and temperature are added, what is the final total volume of the gas.
Vi = 6.0 L and ni = 0.5 moles.
When 0.25 moles are added
nf = ni + 0.25 moles
The final volume of the gas is 9.0 L.
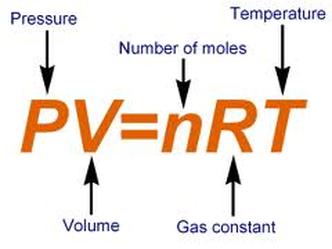
Combined and Ideal gas laws:
The combined gas law is a gas law that combines Charles's law, Boyle's law, and Gay-Lussac's law. There is no official founder for this law;
it is merely an amalgamation of the three previously discovered laws.
Applications:
The combined gas law can be used to explain the mechanics where pressure, temperature, and volume are affected. For example: air conditioners, refrigerators and the formation of clouds.and also use in fluid mechanics and thermodynamics.
By combining (1) and either of (2) or (3), we can gain a new equation with P, V and T. If we divide equation (1) by temperature and multiply equation (2) by pressure we will get:
PV/T = k1(T) / T
PV/T = k2(P) P
Boyle's law:
Boyle's law shows that, at constant temperature, the product of an ideal gas pressure and volume is always constant. It can be determined experimentally using a pressure gauge and a variable volume container. It can also be found through the use of logic; if a container, with a fixed number of molecules inside, is reduced in volume, more molecules will hit the sides of the container per unit time, causing a greater pressure.
As a mathematical equation, Boyle's law is:
P1V1 = P2V2
where P is the pressure (Pa), V the volume (m3) of a gas, and k1 is the constant from this equation—it is not the same as the constants from the other equations below.
This is known as Boyle's law which states: the volume of a given mass of gas is inversely proportional to its pressure, if the temperature remains constant. Mathematically this is:
V= K/P
(k is a constant)
Charle's law:
Charles's Law is an ideal gas law where at constant pressure, the volume of an ideal gas is directly proportional to its absolute temperature.
V1/T1 = Vf/Tf
where:
V1 = initial pressure
T1 = initial temperature
Vf = final pressure
Tf = final temperature
Gay-Lussac's law:
Gives the relationship between pressure and temperature when volume and amount are held constant.
If the temperature of a container is increased, the pressure increases.
If the temperature of a container is decreased, the pressure decreases.
The mathematical form of Gay-Lussac's Law is: P ÷ T = k
This means that the pressure-temperature fraction will always be the same value if the volume and amount remain constant.
Let P1 and T1 be a pressure-temperature pair of data at the start of an experiment. If the temperature is changed to a new value called T2, then the pressure will change to P2.
This equation of P1 ÷ T1 = P2 ÷ T2 will be very helpful in solving Gay-Lussac's Law problems.
Example: 5.00 L of a gas is collected at 22.0°C and 745.0 mmHg. When the temperature is changed to standard, what is the new pressure?
Solution: convert to Kelvin and insert:
745.0 mmHg / 295.0 K = x/ 273.0 K
Cross multiply and divide for the new pressure.
Avogadro's laws:
Avogadro's law is an experimental gas law relating volume of a gas to the amount of substance of gas present. A modern statement of Avogadro's law is:
A law that states, equal volumes of all gases, at the same temperature and pressure, have the same number of molecules.
For a given mass of an ideal gas, the volume and amount (moles) of the gas are directly proportional if the temperature and pressure are constant.
which can be written as:
v/n = K
The most important consequence of Avogadro's law is that the ideal gas constant has the same value for all gases. This means that the constant
P1 x V1 / T1x n1 = P2 x V2 / T2 x n2 = constant
Example:
A 6.0 L sample at 25 °C and 2.00 atm of pressure contains 0.5 moles of a gas. If an additional 0.25 moles of gas at the same pressure and temperature are added, what is the final total volume of the gas.
Vi = 6.0 L and ni = 0.5 moles.
When 0.25 moles are added
nf = ni + 0.25 moles
The final volume of the gas is 9.0 L.
Combined and Ideal gas laws:
The combined gas law is a gas law that combines Charles's law, Boyle's law, and Gay-Lussac's law. There is no official founder for this law;
it is merely an amalgamation of the three previously discovered laws.
Applications:
The combined gas law can be used to explain the mechanics where pressure, temperature, and volume are affected. For example: air conditioners, refrigerators and the formation of clouds.and also use in fluid mechanics and thermodynamics.
By combining (1) and either of (2) or (3), we can gain a new equation with P, V and T. If we divide equation (1) by temperature and multiply equation (2) by pressure we will get:
PV/T = k1(T) / T
PV/T = k2(P) P