PHYSICAL AND CHEMICAL CHANGES
What is the difference between chemical and physical change?
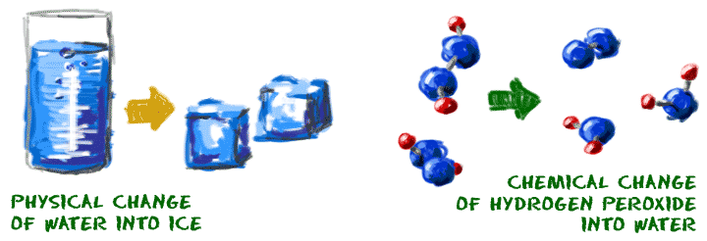
Chemical change is any change that results in the formation of new chemical substances. At the molecular level, chemical change involves making or breaking of bonds between atoms
Looking at Molecules-
Chemical changes happen on a much smaller scale. While some experiments show obvious chemical changes, such as a color change, most chemical changes that happen between molecules are not seen. Melting a sugar cube is a physical change, because the substance is still sugar. Burning a sugar cube is a chemical change.
Chemical change is any change that results in the formation of new chemical substances. At the molecular level, chemical change involves making or breaking of bonds between atoms
- iron rusting (iron oxide forms)
- gasoline burning (water vapor and carbon dioxide form)
- eggs cooking (fluid protein molecules uncoil and crosslink to form a network)
- bread rising (yeast converts carbohydrates into carbon dioxide gas)
- whipping egg whites (air is forced into the fluid, but no new substance is produced.
- boiling water (water molecules are forced away from each other when the liquid changes to vapor, but the molecules are still H2O.)
- dissolving sugar in water (sugar molecules are dispersed within the water, but the individual sugar molecules are unchanged.)
- dicing potatoes (cutting usually separates molecules without changing them.)
Looking at Molecules-
Chemical changes happen on a much smaller scale. While some experiments show obvious chemical changes, such as a color change, most chemical changes that happen between molecules are not seen. Melting a sugar cube is a physical change, because the substance is still sugar. Burning a sugar cube is a chemical change.